
However, some nuclides decay faster than others. To determine the age of a sample whose activity has been measured by beta counting, the ratio of its activity to the activity of the standard must be found.Half-Life and the Rate of Radioactive Decay A secondary standard, Oxalic Acid SRM 4990C, also referred to as HOxII, 1,000 lb of which was prepared by NIST in 1977 from French beet harvests, is now in wide use. Īll of this first standard has long since been consumed, and later standards have been created, each of which has a given ratio to the desired standard activity. This is addressed by defining the standard to be 0.95 times the activity of HOxI. Since it was created after the start of atomic testing, it incorporates bomb carbon, so measured activity is higher than the desired standard. The first standard, Oxalic Acid SRM 4990B, also referred to as HOxI, was a 1,000 lb batch of oxalic acid created in 1955 by the National Institute of Standards and Technology (NIST). These samples contain carbon of a known activity. īoth beta counting and AMS measure standard samples as part of their methodology. The resulting standard value, A abs, is 226 becquerels per kilogram of carbon. The fossil fuel effect was eliminated from the standard value by measuring wood from 1890, and using the radioactive decay equations to determine what the activity would have been at the year of growth. Because of the fossil fuel effect, this is not actually the activity level of wood from 1950 the activity would have been somewhat lower. In order to allow measurements to be converted to the 1950 baseline, a standard activity level is defined for the radioactivity of wood in 1950. 
This convention is necessary in order to keep published radiocarbon results comparable to each other without this convention, a given radiocarbon result would be of no use unless the year it was measured was also known-an age of 500 years published in 2010 would indicate a likely sample date of 1510, for example. Īnother standard is the use of 1950 as "present", in the sense that a calculation that shows that a sample's likely age is 500 years "before present" means that it is likely to have come from about the year 1450. 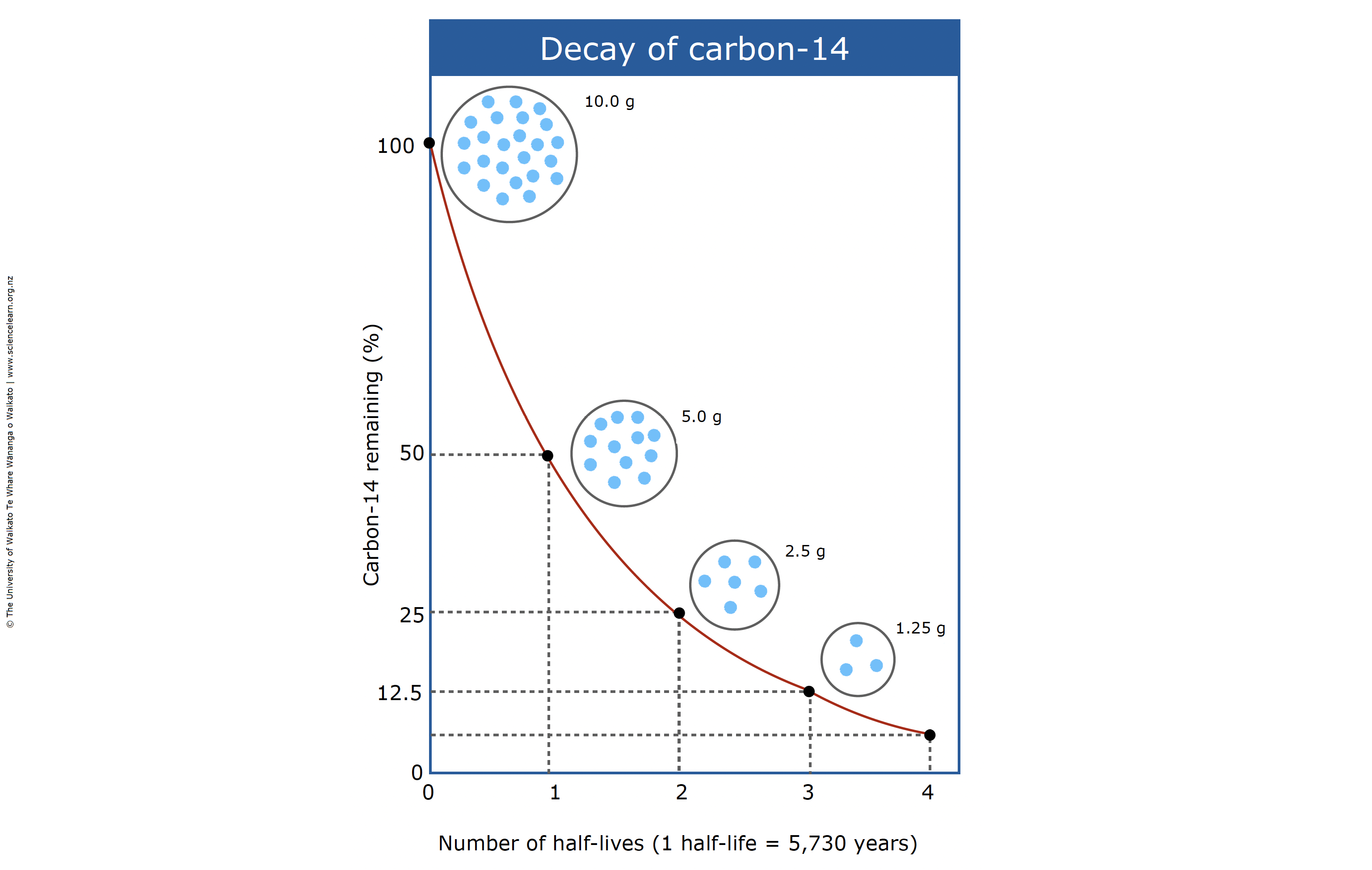
The details of the calculations for beta counting and AMS are given below. This is possible because the δ 13C of wood is known, and the δ 13C of the sample material can be measured, or taken from a table of typical values. To compensate for this, the measurements are converted to the activity, or isotope ratio, that would have been measured if the sample had been made of wood. Since different materials have different δ 13C values, it is possible for two samples of different materials, of the same age, to have different levels of radioactivity and different 14Ĭ ratios. A related standard is the use of wood, which has a δ 13C of -25‰, as the material for which radiocarbon ages are calibrated. One of these, the standard for normalizing δ 13C values, is Pee Dee Belemnite (PDB), a fossil which has a 13Ĭ ratio of 1.12372%. The calculations to convert measured data to an estimate of the age of the sample require the use of several standards. The calculations to be performed depend on the measurements taken based on the technology used, since beta counters measure the sample's radioactivity, whereas accelerator mass spectrometers (AMS) determine the ratio of the three different carbon isotopes in the sample. There are several possible sources of error in both the beta counting and AMS methods. Calculations of radiocarbon dates are typically made based on measurements from beta counting devices or from accelerator mass spectrometers (AMS). Radiocarbon dating is also referred to as carbon dating or carbon-14 dating. Radiocarbon dating methods produce data based on the ratios of different carbon isotopes in a sample that must then be further manipulated in order to calculate a resulting "radiocarbon age". The calculation of radiocarbon dates determines the age of an object containing organic material by using the properties of radiocarbon (also known as carbon-14), a radioactive isotope of carbon. Please consider expanding the lead to provide an accessible overview of all important aspects of the article. This article's lead section may be too short to adequately summarize the key points.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
